The molecular mechanisms of visual chromophore release from cellular retinaldehyde-binding protein
Abstract
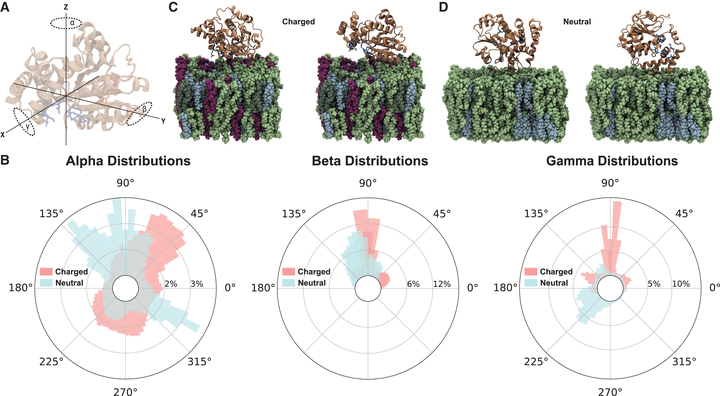
Cellular retinaldehyde-binding protein (CRALBP) is an 11-cis-retinoid binding protein operating within the visual cycle. CRALBP serves as the terminal acceptor of 11-cis-retinaldehyde (11cRAL) produced within the retinal pigment epithelium (RPE) and mediates 11cRAL transport to the RPE apical microvilli. Crystallographic structures of CRALBP revealed that the 11cRAL-binding pocket is sealed off from bulk solvent, indicating a necessity for conformational changes to allow ligand egress. Here, we performed long timescale all-atom molecular dynamics simulations of CRALBP to elucidate the mechanisms of ligand release. CRALBP exhibits slower diffusive behavior in the presence of membranes containing negatively charged phospholipids, which bind to an exposed cationic pocket in CRALBP. Umbrella sampling calculations revealed thermodynamically likely pathways for 11cRAL egress. Our data suggest that the CRALBP-acidic phospholipid interaction facilitates 11cRAL release through allosteric, conformational changes that perturb the binding site, lowering ligand affinity. These findings offer insights into the molecular pathology of CRALBP-associated retinopathy.